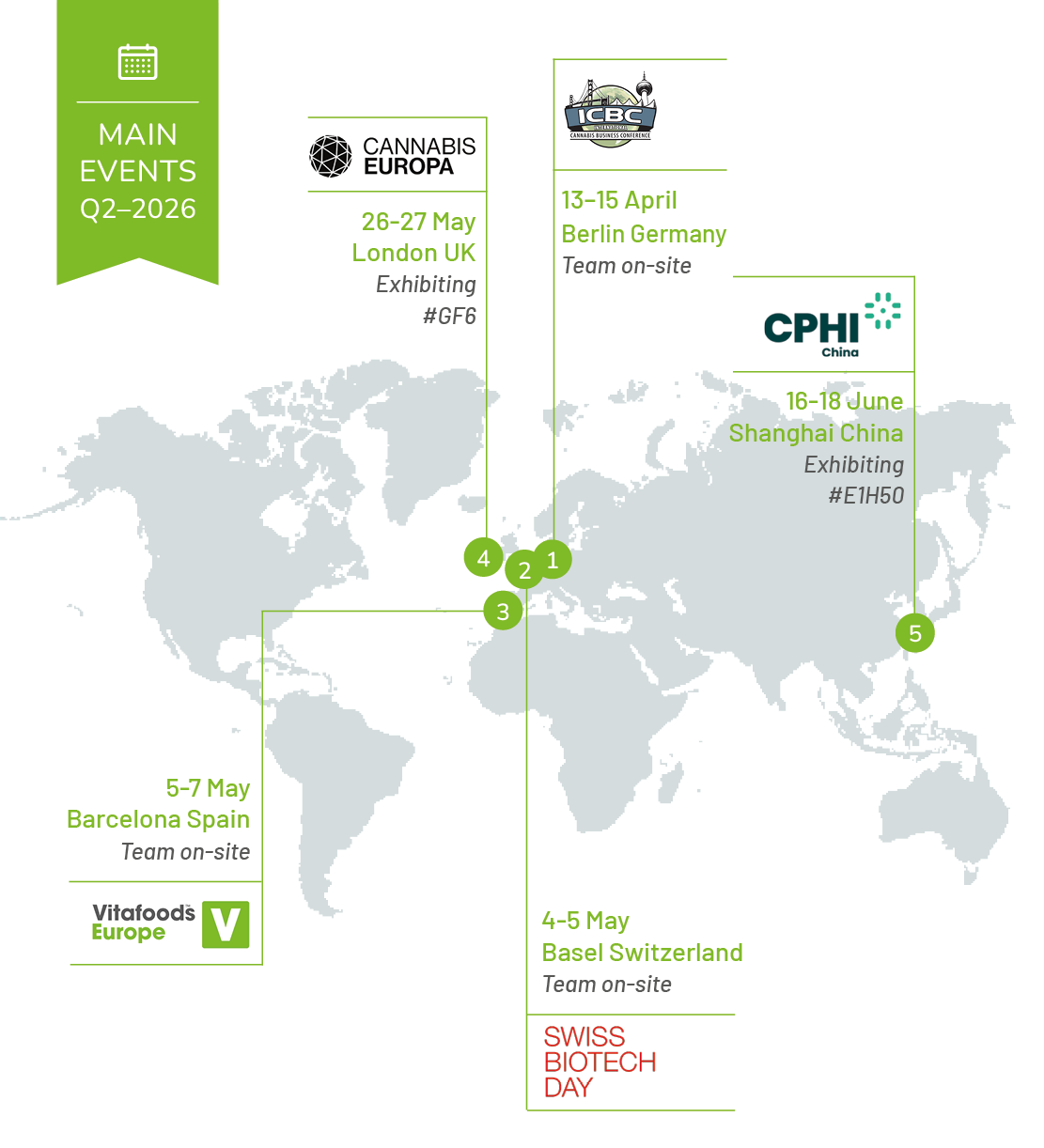
APRIL

13-15 April
ICBC International Cannabis Business Conference
Berlin, Germany
Estrel Berlin Hotel
> Visit conference web page